Solving the Silent Mass Disaster: The Role of Medical Implants in Forensic Identification
Dr. Shabanna Smith, DFS
Oklahoma State University
Abstract:
Every year approximately 4,400 unidentified bodies are discovered. The forensic community has made significant technological advancements—utilizing tools such as dental molds, DNA analysis, fingerprinting, and genetic genealogy to assist in postmortem investigations. Despite these advancements, the issue remains a significant challenge, often described by experts as “a mass disaster over time.” This phrase powerfully conveys the ongoing nature of the problem and the urgency with which it needs to be addressed. Due to our aging human population and increased numbers of surgical procedures being performed, there is a growing number of biomedical devices being implanted every year. This study aims to highlight the significance of unidentified human remains and how they can be positively identified via medical implant markings. It is important to understand how these advancements not only build upon past technologies but require more research to be fully effective. When implants are regulated and properly marked, they can prove beyond useful in forensic investigations. Personnel at numerous national forensic offices confirmed the value of medical implants. They added that orthopedic implants are most common2.
Keywords: Missing persons; Unidentified human remains; Postmortem investigations; Forensic community; Implants; Medical devices; Medical Implants; Solve crimes; Forensic tool Consistent with perspectives advanced in biomedical engineering, medical device technology is inherently interdisciplinary10.
Introduction
When reflecting on our nation’s most devastating mass disasters—many Americans recall the 9/11 attacks, the aftermath of Hurricane Katrina, or the global effects of the COVID-19 pandemic (Avraham, 2020). As time progresses, mass disasters continue to occur, and we are left with the consequences. These events have left their irrefutable marks on society and have contributed to an uptick in missing people and unidentified human remains cases. While these disasters are devastating, they have also contributed to a quieter, ongoing crisis that persists year after year, in morgues and forensic labs across the country. Missing persons and unidentified remains present some of the most complex obstacles for law enforcement, forensic investigators, and medical examiners. According to the FBI’s National Crime Information Center, an average of over 600,000 individuals were reported missing annually in the United States from 2007-2020. As of May 2025, approximately 15,000 active unidentified persons' cases remain unsolved. These staggering figures underscore a persistent and deeply troubling issue, one that experts have termed “a mass disaster over time”1. Given the scale of missing persons' cases and the limitations of traditional identification methods, the forensic community has sought innovative approaches to address this persistent challenge. As Dr. Richard Kimble says in The Fugitive (1993), “I'm trying to solve a puzzle.” This quote encapsulates the investigative nature of forensic science, where professionals must piece together fragmented evidence, often including medical implants—to identify the unknown and bring resolution to families.
Background and Context
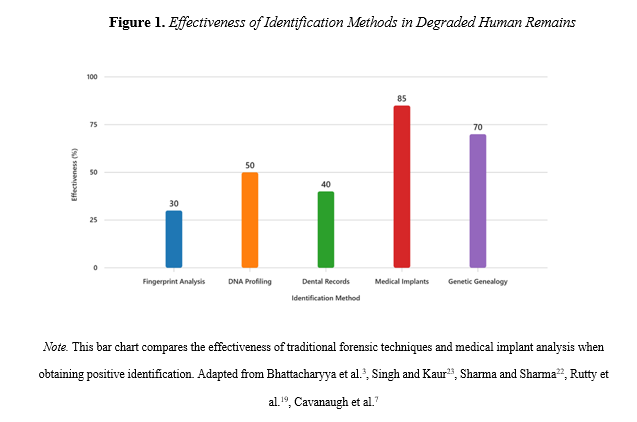
In response to this ongoing crisis, the forensic science community has made strides in their technological advancements. Traditional tools such as dental molds, DNA profiling, genetic genealogy, and fingerprint analysis have become essential in achieving positive identification during postmortem investigations (Figure 1). However, these methods can often be recognized as ineffective, especially when human remains are degraded, fragmented, or exposed to other extreme conditions which are quite common in mass disasters21. These methods might also not be used due to ethical constraints and accuracy. For example, genealogical DNA testing can raise privacy concerns, especially when using public databases. Medical device analysis avoids these issues by relying on existing medical records and manufacturer data.
Furthermore, a study was conducted to assess the accuracy and reliability of forensic latent fingerprint decisions25. The article stated in general that the examiners frequently differed on whether fingerprints were suitable for reaching a conclusion. The understanding of forensic fingerprint evidence relies on the expertise of a latent print examiner. The accuracy of decisions made by latent print examiners has not been established in a large setting, despite the forensic applicability over the past hundred years. Previous studies show that there is an increase in scrutiny of the discipline resulting from errors and a series of court admissibility challenged to the scientific basis of fingerprint evidence25. In these cases, forensic investigators must turn to alternative techniques to solve missing persons, and unidentified human remains cases. So, despite some of these advancements (dental molds, DNA profiling, genetic genealogy, and fingerprint analysis), the problem persists because the forensic community must rely on a culmination of all the techniques to achieve positive identification.
In recent years, medical implant investigation has emerged as a valuable tool for forensic identification. It can be supplemental in solving unidentified human remains cases. Throughout this study, we will explore how regulated medical implants can supplement existing technologies in postmortem investigations. The key research question is- how can medical implants provide valuable information to forensic experts? It is important to note that medical devices and/or implants are more prevalent today because of the aging global population, the rise of chronic diseases, and advancements in technology. Since there is an increase in use—upon recovery of unidentified remains, forensic pathologists can explant the implant at autopsy and examine to help solve forensic investigations. To do so, they must do the initial recovery and examination. This is where the scene's documentation takes place; the location is recorded as well as the condition of the remains. A visual inspection should be conducted. Signs of medical implants (i.e. metal plates, screws, prosthetics) prove fruitful. Also, postmortem radiographs can be used to detect internal implants that are not visible externally. By carefully extracting implants, detailed documentation can begin—type of implant, manufacturer’s logo and name, serial number and lot number. By contacting the manufacturer, the serial/lot number can be used to trace the implant distribution.
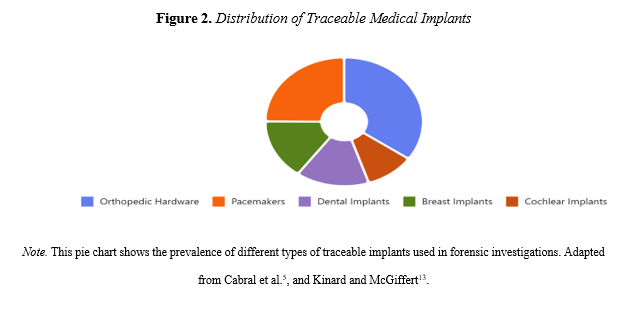
Furthermore, the manufacturer can provide guidance on requesting implant tracking data per FDA’s Safe Medical Devices Act of 1990 and Modernization Act of 1997. From this information, hospital and surgeon details, who performed the implantation, can be traced, with legal authorization. This can be used to match implant data with known cases. Demographic clues (age, location of surgery) can be used to narrow down candidates. The serialization of medical implants is governed by the Unique Device Identification (UDI) system, which is mandated by regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Union's Medical Device Regulation (MDR)27. Once an implant is serialized, manufacturers record the UDI in centralized databases. Healthcare Providers document the UDI in the patient's medical records. Patients may receive implant identification cards containing the UDI and relevant device information. This multi-tiered documentation ensures traceability from the point of manufacture to implantation27. These devices range from orthopedic hardware and pacemakers to breast augmentation devices (Figure 2).
They are designed to withstand biological and environmental stressors. To review, many implants contain serial numbers, manufacturer logos, and material compositions that can be traced back to patient records, healthcare providers, or manufacturers. These markers or identifiers (i.e. serial numbers and manufacturer logos) can be critical when solving crimes. Especially when the medical implants, which the markers are adhered to, are properly documented and regulated. They can be matched with medical records, hospital databases, or manufacturer registries to establish a link between the implant and its host. This process can significantly narrow the suspect pool and aid in positive identification. This is helpful in situations where traditional methods are not applicable.
Many implants are designed to withstand extreme temperatures. They often remain intact even after other biological evidence has deteriorated. This robustness makes them useful in cases where there are victims or cases with extreme weather conditions. As forensic science continues to grow, the collaboration of medical implant analysis into standard post death procedures could enhance identification efforts and significantly reduce the number of unresolved cases. This study aims to evaluate the effectiveness of implant tracking systems and explore how regulatory improvements and technological advancements could further support forensic investigations. Forensic scientists tend to lean on more non-traditional methods, such as medical device investigation. Medical device investigation can withstand the passage of time and environmental exposure. A convincing example happened in 2024, when forensic anthropologists investigated partially burned human remains found in a backyard fire pit. Despite extensive thermal damage, an orthopedic implant in the lower leg remained intact. Using radiographic imaging and serial number tracing, investigators linked the implant to surgical records, leading to a positive identification of the decedent 15 (Figure 3). This case underscores the forensic value of medical implants, especially in scenarios where other identifiers are destroyed. This research explores the forensic potential of medical implant markings in the identification of human remains. By examining existing literature and proposing a survey to forensic professionals, this study aims to assess current practices, identify barriers, and propose recommendations for integrating implant data into routine forensic workflows.

Examining Existing Literature
Medical implants have increasingly become a valuable tool in forensic investigations, offering both physiological data and physical identifiers that can aid in postmortem identification. The literature reveals two primary forensic applications: device interrogation for physiological data and implant tracking through serial numbers and manufacturer records.
Device Interrogation for Time and Cause of Death
Yael Maxwell (2017), a senior medical journalist for TCTMD, highlights the potential of cardiac device interrogation in forensic medicine. She notes that approximately 30% of forensic cases remain unresolved due to unclear cause or time of death. In her study of 150 autopsied patients with cardiac implants, traditional forensic methods failed to determine time of death in about 25% of cases. However, device interrogation successfully pinpointed the time of death in 36.8% of those cases. Maxwell emphasizes that while cardiologists are not coroners, postmortem interrogation of cardiac devices should be routinely performed to aid in determining timing, mechanism, and cause of death14. A compelling case reported by Dobson (2003) further illustrates this potential. A 72-year-old man with a dual- chamber pacemaker was found beaten to death outside his home. Despite severe trauma, the pacemaker remained functional and provided telemetric data that reconstructed the victim’s final hours—including when he awoke, walked, encountered the intruder, and died. This case demonstrates how cardiac devices can supplement traditional forensic methods when establishing a timeline of death8. Throughout the literature review, several other case studies came to light that further supported thesis. The few cases detailed above only account for a small percentage of the documented cases where MedTech devices lead to positive identity.
Implant Markings for Positive Identification
Beyond physiological data, the physical characteristics of implants—such as serial numbers, manufacturer logos, and material composition offer a powerful avenue for identifying human remains. Regulatory frameworks like the Unique Device Identification (UDI) system, established by the FDA, assign standardized codes to medical implants, enabling forensic experts to trace devices back to surgical records, healthcare providers, or manufacturers26. Dental implants are particularly useful due to their durability and traceability. Sivaswamy and Vasudevan (2023) highlight their resilience to fire, decomposition, and trauma, making them reliable forensic markers24. Cattaneo et al. (2006) describe a case in which skeletal remains were identified through a dental implant matched to antemortem records, demonstrating the implant’s forensic value. Orthopedic implants also show promise 6. Bukhamseen et al. (2022) conducted a retrospective study of 608 forensic autopsies in the U.S., where 56 cases required alternative identification methods due to decomposition, blunt force trauma, or charring. Of those, 8 had known implants, and 4 were successfully identified using serial numbers on the surgical devices4. Throughout all of the literature examined, orthopedic implants seemed to be the most common for bearing lot numbers or unique identifiers leading to positive identification.
Case Studies Demonstrating Practical Application
After reviewing literature, understanding how medical devices can be used may be better followed with practical application. Jaiswani et al. (2017) reports a case involving a charred body found in a remote area. The deceased, an adult female, had a prosthetic implant bearing lot and serial numbers12. Investigators traced the implant to a local distributor, who identified the manufacturer. The manufacturer’s database linked the device to a specific doctor, who then provided the patient’s identity. Although the process was complex, it proved effective in a case where traditional identification methods were not viable. Building on these documented cases and theoretical applications, this study proposed a survey to explore how forensic professionals currently engage with medical implant data in real-world settings. These examples underscore the practicality of implant-based identification, especially in cases involving decomposition, mutilation, burial, or cremation. While the process may be drawn out, it offers a viable solution when conventional forensic techniques fall short.
Survey of Forensic Professionals
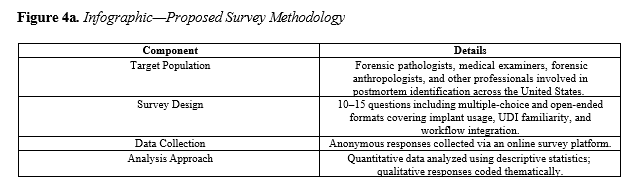
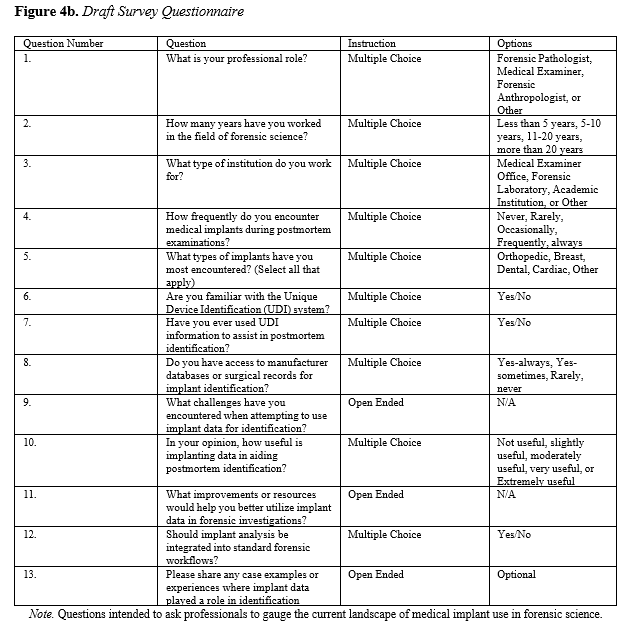
Although a formal survey was not conducted due to time constraints, this study proposes a structured survey as a future research method to assess the practical use of medical implants in forensic identification. The goal of the proposed survey is to gather insights from forensic professionals regarding their experiences, challenges, and perspectives on incorporating implant data into routine postmortem investigations. The survey would be distributed to forensic pathologists, medical examiners, forensic anthropologists, and other professionals involved in postmortem identification. Participants would be selected from medical examiner offices, forensic laboratories, and academic institutions across the United States. The proposed survey would consist of approximately 10–15 questions, including both multiple-choice and open-ended formats. Key topics would include frequency of encountering medical implants during autopsy, familiarity with the Unique Device Identification (UDI) system, access to manufacturer databases or surgical records, challenges in using implant data for identification, and opinions on integrating implant analysis into standard forensic workflows. Responses would be collected anonymously using an online survey platform. Quantitative data would be analyzed using descriptive statistics to identify trends, while qualitative responses would be coded thematically to highlight common barriers and recommendations (Figure 4a and 4b). This proposed methodology is intended to complement the literature review and case studies presented in this paper. While the proposed survey outlines a future direction for data collection, it is also important to examine existing regulatory frameworks and forensic workflows that shape current practices. Building on the proposed survey, it is essential to understand the current landscape of medical implant use in forensic science. This includes examining the regulatory systems, technological features, and quality controls that make implants viable tools for identification.
Assessing Current Practices
Medical implants are increasingly recognized as valuable forensic tools, not only because of their traceability but also due to the rigorous regulatory and quality systems that govern their design, manufacture, and documentation. Understanding current practices in the medical device industry helps clarify why implants are reliable identifiers in postmortem investigations. Before a medical device can be marketed and implanted, it must undergo a stringent regulatory approval process. In the United States, the Food and Drug Administration (FDA) oversees this process, requiring manufacturers to demonstrate safety, effectiveness, and biocompatibility20. Similar regulatory bodies exist globally, such as the National Medical Products Administration (NMPA) in China, the European Medicines Agency (EMA) in Europe, and the Gulf Health Council in the Middle East11. These agencies enforce standards that ensure implants are traceable, durable, and manufactured under controlled conditions—making them suitable for forensic use even years after implantation.
One of the most impactful regulatory developments in recent years is the implementation of the Unique Device Identification (UDI) system. The UDI assigns a standardized code to each medical device, which includes information about the manufacturer, model, and production batch. This code is typically etched or embedded on the device itself and linked to a global database accessible to healthcare providers and regulators. In forensic investigations, UDIs allow examiners to trace implants back to surgical records, manufacturers, or healthcare facilities, significantly narrowing the pool of potential identities 27, 9.
However, it is important to note that there are regions where medical devices are manufactured and implanted without adherence to international regulatory standards. In countries with limited oversight—such as parts of Latin America, including Mexico—unregulated implants have been linked to severe complications and fatalities. These devices often lack traceable identifiers, making postmortem identification nearly impossible and posing significant public health risks.
Reports indicate that counterfeit or substandard implants, including orthopedic and cosmetic devices, enter the market through informal supply chains, bypassing quality checks and sterilization requirements 28,18. The absence of UDI systems in these regions not only undermines patient safety but also complicates forensic investigations, as examiners cannot rely on device markings to establish identity or cause of death.
Medical implants are engineered to withstand biological and environmental stressors. Materials such as titanium, stainless steel, and medical-grade polymers are chosen for their durability, corrosion resistance, and thermal stability. These properties make implants resilient in scenarios involving decomposition, fire, or prolonged burial. Forensic case studies have shown that implants often remain intact even when other biological identifiers are destroyed, making them a reliable source of postmortem data16. Quality assurance (QA) systems play a critical role in ensuring the consistency and traceability of medical implants. Manufacturers are required to maintain detailed documentation throughout the product lifecycle, including design specifications, production records, and distribution logs. These QA systems are audited regularly by regulatory agencies and are essential for forensic investigators seeking to verify implant origin and authenticity17. When properly maintained, these records can be used to match an implant to a specific patient, even in the absence of traditional identifiers like fingerprints or DNA.
Confirming Current Practices with Professionals in the Field
To validate the accuracy of the practices described in this section, informal outreach was conducted with multiple medical examiners and coroner offices across different jurisdictions, including offices in Minnesota, Texas, and South Carolina. Additionally, a conversation was held with David Leavitt, CEO of American Forensics, a nationally accredited independent forensic service provider. All respondents confirmed that the information presented—such as the reliance on dental records, fingerprints, and DNA as primary identification methods—accurately reflects current procedures. Importantly, every office contacted reported that when a medical implant is present, implant analysis is consistently utilized as part of the identification process. Respondents emphasized that serial numbers or manufacturer markings on implants are checked against surgical records or manufacturer databases whenever possible, and this practice is considered standard whenever such identifiers are available. While systematic integration into national databases remains limited, these confirmations underscore that implant analysis is already recognized as a valuable tool in forensic identification2. This finding supports the argument that formalizing and expanding these practices could significantly enhance identification outcomes for missing persons and unidentified remains.
Barriers to Using Medical Implants for Forensic Identification
Despite the growing potential of medical implants in forensic science, several barriers limit their routine use in postmortem identification. These challenges stem from gaps in software/systems/infrastructure, regulation, and resource availability.
Lack of a Centralized Tracking Database
One of the most significant obstacles is the absence of a centralized, publicly accessible database that links implant serial numbers to patient identities. While systems like the FDA’s AccessGUDID provide manufacturer and device information, they do not connect implants to individual patients. This limitation forces forensic investigators to rely on indirect methods—such as contacting hospitals or manufacturers—which can be time-consuming and may not yield results, especially in older cases or when records are incomplete.
Unregulated or Poorly Documented Implants
Not all medical implants are subject to the same level of regulatory oversight. Devices implanted outside of major regulatory jurisdictions—such as in countries with limited medical device governance—may lack standardized identifiers or traceable documentation. Additionally, implants used in cosmetic or elective procedures may not be recorded in centralized systems, making them difficult to trace. This lack of uniformity complicates efforts to use implant data for forensic identification. From a regulatory toxicologist’s perspective, the challenge is compounded by variability in biocompatibility testing standards. The ISO 10993 series, which governs biological evaluation of medical devices, provides a framework for assessing the safety of materials used in implants. However, not all devices, especially those used in non-therapeutic or short-term applications, undergo comprehensive testing across the full battery of endpoints (e.g., cytotoxicity, sensitization, genotoxicity, systemic toxicity). Moreover, healthcare-use devices (e.g., catheters, wound dressings) are often designed for short-term or transient contact and may not require the same level of traceability or durability as long-term implantable devices (e.g., orthopedic screws, pacemakers, neurostimulators). Long-term implants typically undergo more rigorous documentation and serialization, making them more useful in forensic identification. In contrast, poorly documented implants, especially those not intended for permanent use—may lack unique identifiers, manufacturer records, or compatibility with global device registries. This disparity in regulatory rigor and documentation standards underscores the need for forensic protocols that account for the biological and regulatory context of the implant, not just its physical presence.
Limited Resources for Tracing Lot Numbers
Even when implants are properly labeled with serial or lot numbers, tracing them back to a specific patient can be resource intensive. Investigators may need to contact multiple entities—such as distributors, manufacturers, and healthcare providers—to reconstruct the implant’s history. Smaller jurisdictions or underfunded forensic laboratories often lack the personnel, time, or access required to pursue these leads effectively. Without streamlined workflows or automated systems, the process can become prohibitively slow or incomplete.
This challenge is particularly pronounced in cold cases, where the passage of time further complicates access to records and institutional memory. Medical device data that could potentially identify a decedent may remain uninvestigated for years due to staffing shortages or lack of specialized training. In some cases, implants with identifiable markings are recovered, but the absence of a centralized database or coordinated inter-agency support means the information is never fully utilized. As a result, cases remain open not because evidence is lacking, but because the infrastructure to act on that evidence is insufficient.
While these barriers present real challenges, emerging technologies and collaborative frameworks offer promising solutions. As Yael notes, using medical implant evidence— whether data or physical markings—outside of a clinical setting is not unprecedented. Therefore, concerns about using medical devices beyond their intended clinical purpose may be unfounded14. Integrating medical device traceability into forensic workflows could significantly reduce the backlog of unidentified remains and accelerate resolution in long-standing cases.
Future Integration and Technology
As forensic science continues to evolve, the integration of medical implant data into routine postmortem workflows presents a promising future. To fully realize the potential of implants in forensic identification, future efforts must focus on technological innovation, data accessibility, and procedural standardization.
Artificial Intelligence and Automated Workflows
Artificial intelligence (AI) offers powerful tools for streamlining implant identification. Machine learning algorithms can be trained to recognize implant types, manufacturer logos, and serial numbers from radiographic images or autopsy photographs. These systems could automatically implant data with manufacturer databases or hospital records, significantly reducing the time and manual effort required for identification. AI-driven platforms may also assist in pattern recognition across cases, helping forensic teams identify trends in implant use and traceability. This workflow directly addresses previously discussed barriers to implant traceability.
Centralized Implant Databases
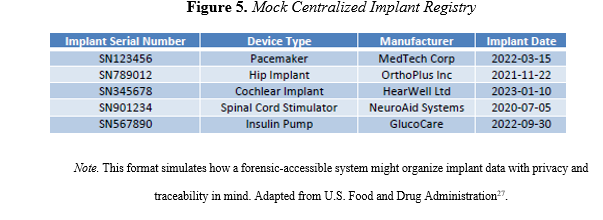
One of the most critical advancements needed is the development of a centralized, secure database that links implant serial numbers to patient records. While systems like the FDA’s AccessGUDID provide device-level information, they do not connect implants to individual patients. A forensic-accessible registry—ideally with tiered privacy protections— could allow authorized personnel to trace implants directly to surgical histories, improving identification accuracy in cases where traditional methods fail (Figure 5). This provides a real solution for one of the hindering barriers that investigators and those alike observe. Structured data could support forensic investigations by enabling authorized personnel to trace implants back to surgical histories while maintaining patient confidentiality.
Integration into Autopsy Protocols
To make implant analysis a routine part of forensic practice, standardized protocols must be developed and adopted across jurisdictions. These protocols should include guidelines for explanting devices, documenting serial numbers, and submitting data for cross- referencing. Training programs for medical examiners and forensic pathologists could ensure consistent handling and reporting of implant evidence. Additionally, collaboration with healthcare systems and device manufacturers will be essential to streamline access to implant records.
Conclusion
Taken together—the literature, proposed methodology, and current practices—highlight a growing opportunity to enhance forensic identification through medical implant analysis. As echoed in pop culture by Dr. Richard Kimble in The Fugitive (1993), “I'm trying to solve a puzzle.” This quote reflects the reality faced by forensic professionals who must navigate complex evidence trails— often relying on medical implants—to bring resolution to silent mass disasters.
This study found that traditional forensic methods such as fingerprinting and DNA analysis are often ineffective in cases involving decomposition, fragmentation, or thermal damage. Medical implants, particularly orthopedic and dental devices, offer durable, traceable identifiers that can survive extreme conditions and aid in postmortem identification. Case studies demonstrate successful identification using implant serial numbers and manufacturer records, even when other biological evidence was compromised.
Regulatory systems like the FDA’s Unique Device Identification (UDI) framework provide a foundation for traceability, though gaps remain in linking devices to individual patients. Barriers such as lack of centralized databases, inconsistent documentation, and limited forensic resources hinder widespread adoption of implant-based identification.
Future integration of AI, centralized registries, and standardized autopsy protocols could streamline workflows and improve identification outcomes. In conclusion, while traditional forensic techniques remain foundational, the integration of medical implant data offers a powerful supplement—especially in cases where other identifiers are compromised. By addressing regulatory gaps, investing in centralized databases, and leveraging AI, the forensic community can significantly improve identification outcomes and bring closure to families affected by unresolved cases. This approach offers a promising solution to the persistent crisis of unidentified human remains—what experts have termed the “silent mass disaster."
References
1. Ambers, A. (2023). Missing persons and unidentified human remains: The world’s silent mass disaster. In Forensic Genetic Approaches for Identification of Human Skeletal Remains (pp. 1–14). Academic Press. https://doi.org/10.1016/B978-0-12-815766- 4.00001-7
2. American Forensics. (n.d.). About us. Retrieved from https://www.americanforensics.com Avraham, E. (2020). From 9/11 through Katrina to Covid-19: crisis recovery campaigns for American destinations. Current Issues in Tourism, 24(20), 2875–2889. https://doi.org/10.1080/13683500.2020.1849052
3. Bhattacharyya, D., Ranjan, R., & Kim, T.-h. (2023). A review of methods employed for forensic human identification. In Advanced Biometric Technologies (pp. 1–25). Springer. https://doi.org/10.1007/978-3-031-45952-8_1
4. Bukhamseen, A. H., Aldhameen, A. A., Alzayat, N. T., Alqadeeb, M. M., Alotaibi, B.
K., Aljohani, R. I., & Menezes, R. G. (2022). The use of orthopedic surgical implants for forensic identification: An overview. Acta Biomedica, 93(3), e2022082. https://doi.org/10.23750/abm.v93i3.1247
5. Cabral, J. A., Machado, D. A., Bridi, A. C., Marta, C. B., Silva, P. A. L., Alves Junior, J. L.,
... & Neves, R. B. (2021). The traceability of implantable medical devices and the unique device identification system: A bibliometric study. Global Academic Nursing Journal, 2(2). https://dx.doi.org/10.5935/2675-5602.20200200
6. Cattaneo, C., De Angelis, D., Porta, D., & Grandi, M. (2006). Personal identification of cadavers and human remains. In A. Schmitt, E. Cunha, & J. Pinheiro (Eds.), Forensic Anthropology and Medicine (pp. 359–379). Humana Press. https://doi.org/10.1007/978-1-59745-099-7_15
7. Cavanaugh, S., Bowers, M., Bever, R., Byrne, M., Plaza, D., & Davoren, J. M. (2024).
Comparative evaluation of genotyping technologies for investigative genetic
genealogy in sexual assault casework. National Institute of Justice. https://nij.ojp.gov/library/publications/comparative-evaluation-genotyping- technologies-investigative-genetic-genealogy
8. Dobson, R. (2003, January 11). Pacemaker pinpoints time of death in murder case. BMJ, 326(7380), 70. https://doi.org/10.1136/bmj.326.7380.70/a
9. Dhruva, S. S., Ross, J. S., & Wilson, N. A. (2023). Unique Device Identifiers for Medical Devices at 10 Years. JAMA Internal Medicine, 183(10), 1045–1046. https://doi.org/10.1001/jamainternmed.2023.3572
10. Gohar F, et al. Driving medical innovation through interdisciplinarity: unique opportunities and challenges. Frontiers in Medicine, 2019. DOI: 10.3389 fmed.2019.00035
11. Gupte, T., Nitave, T., & Gobburu, J. (2025). Regulatory landscape of accelerated approval
pathways for medical devices in the United States and the European Union. Frontiers in
Medical Technology, 7. https://doi.org/10.3389/fmedt.2025.1586070
12. Jaiswani, A. K., Dere, R., Kukde, H., Sabale, M., & Savardekar, R. R. (2017). Metal implant– A hidden clue to identification: A case report. Indian Journal of Forensic Medicine & Pathology, 10(3), 63–66. https://doi.org/10.5958/0974-0848.2017.00063.X [journals.sagepub.com]
13. Kinard, M., & McGiffert, L. (2021). Medical device tracking—how it is and how it should be. JAMA Internal Medicine, 181(3). https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/2774431
14. Maxwell, Y. L. (2017, June 26). Implanted devices may help determine time and cause of death when autopsy falls short. TCTMD. https://www.tctmd.com/news/implanted- devices-may-help-determine-time-and-cause-death-when-autopsy-falls-short
15. Mercyhurst University Forensic Anthropology Team. (2024). Identification of thermally altered human remains using orthopedic implants: A case study. Forensic Sciences Research. https://doi.org/10.1080/20961790.2024.xxxxxx
16. MedTechNews. (2025). A Comprehensive Review of Medical Implants: Materials, Failure Modes, Regulation, and Future Directions. https://medtechnews.uk/research- reports/a-comprehensive-review-of-medical-implants-materials-failure-modes- regulation-and-future-directions/
17. Pacheco, D. A. J., Bonato, S. V., & Linck, W. (2025). Advancing quality management in the medical devices industry: strategies for effective ISO 13485 implementation. International Journal for Quality in Health Care, 37(1). https://doi.org/10.1093/intqhc/mzaf004
18. Pan American Health Organization. (2022). Health risks of unregulated medical devices in Latin America. Washington, DC: PAHO. Retrieved from https://www.paho.org/en/publications
19. Rutty, G. N., Morgan, B., & Robinson, C. (2023). An interdisciplinary forensic approach to human identification. Wiley Interdisciplinary Reviews: Forensic Science, 5(2), e1484. https://doi.org/10.1002/wfs2.1484
20. Sastry, A. (2014). Overview of the US FDA Medical Device Approval Process. Current Cardiology Reports, 16(494). https://doi.org/10.1007/s11886-014-0494-3
21. Sharma, R., & Gupta, A. (2021). Challenges in forensic identification: A review of traditional and emerging techniques. Egyptian Journal of Forensic Sciences, 11(1), 1–10. https://doi.org/10.1186/s41935-021-00245-3
22. Sharma, R., & Sharma, S. (2023). Dental profiling & advancements in forensic odontology.
IOSR Journal of Dental and Medical Sciences, 23(7), 46–58. https://www.iosrjournals.org/iosr-jdms/papers/Vol23-issue7/Ser-12/G2307124658.pdf
23. Singh, R., & Kaur, M. (2022). DNA profiling in forensic dentistry. Dental Journal of Advance Studies, 10(1), 30–35. https://djas.org/doi/DJAS/pdf/10.5005/djas-11014-0030
24. Sivaswamy, V., & Vasudevan, S. (2023). Dental implants: An overview. In Forensic Odontology: Principles and Practice (pp. 153–162). Springer. https://www.ncbi.nlm.nih.gov/books/NBK615900
25. Ulery, B. T., Hicklin, R. A., Buscaglia, J., & Roberts, M. A. (2011). Accuracy and Reliability of Forensic Latent Fingerprint Decisions. Proceedings of the National Academy of Sciences, 108(19), 7733–7738. https://doi.org/10.1073/pnas.1018707108
26. U.S. Food and Drug Administration. (2018). AccessGUDID (for the public). https://www.fda.gov/medical-devices/global-unique-device-identification-database- gudid/accessgudid-public
27. U.S. Food and Drug Administration. (2022). Unique Device Identification System. https://www.fda.gov/medical-devices/device-advice-comprehensive- regulatory-assistance/unique-device-identification-system-udi-system
28. World Health Organization. (2021). Medical devices: Regulatory systems strengthening. Geneva:





