Inflammatory Signaling via Extracellular Vesicles Following IL-13 Stimulation in Airway Epithelial Cells
Sahil Patel , Jenks High School, Tulsa, OK 74037, USA
Bhuvanesh Kumar Raju., Department of Anatomy and Cell Biology, Oklahoma State University, Center for Health Sciences, Tulsa, OK 74107, USA.
Radhika D. Pande., Department of Biochemistry and Microbiology, Oklahoma State University, Center for Health Sciences, Tulsa, OK 74107, USA.
Vikramsingh Gujar., Department of Anatomy and Cell Biology, Oklahoma State University, Center for Health Sciences, Tulsa, OK 74107, USA.
Correspondence: Vikramsingh Gujar
Email - vikram.gujar@okstate.edu
Address – E-415, 1111 W 17th St Tulsa, OK 74107
FUNDING SOURCE
This study was supported by Startup funds awarded to VG by the Oklahoma State University Center for Health Sciences.
CONFLICT OF INTEREST
The author declares no conflicts of interest related to this study.
ABSTRACT
Background: Extracellular vesicles (EVs) are membrane-bound nanoparticles released by cells that facilitate intercellular communication through the transfer of proteins, lipids, and nucleic acids. Growing evidence implicates EVs in inflammatory signaling; however, their role in type 2 airway inflammation remains poorly characterized. Interleukin-13 (IL-13) is a central cytokine in allergic airway diseases, including allergic rhinitis. This study investigated whether IL-13 stimulation alters EV cargo in airway epithelial cells, thereby contributing to the propagation of inflammatory signals.
Methods: Human airway epithelial A549 cells were cultured and treated with IL-13 for 24 hours, alongside vehicle-treated and untreated control groups. Small EVs were isolated from conditioned media using ExoQuick precipitation. EV yield and integrity were validated by nanoparticle tracking analysis (NTA) and Western blotting for EV-associated markers. The inflammatory chemokine CCL-26 was quantified at the protein level in EVs using Western blotting, while corresponding cellular mRNA expression was assessed by quantitative real-time PCR (qPCR).
Results: Successful isolation of EVs from A549 cells was confirmed by NTA and Western blot analysis. EVs derived from IL-13-treated cells demonstrated a significant increase in CCL-26 protein levels compared with EVs from control and vehicle-treated groups. In contrast, qPCR analysis revealed no significant differences in CCL-26 mRNA expression among the experimental groups, as indicated by comparable Ct values.
Conclusion: IL-13 stimulation selectively enhances the enrichment of CCL-26 protein within extracellular vesicles without corresponding changes in mRNA expression, suggesting post-transcriptional regulation or selective EV cargo loading. These findings support a role for EVs in mediating inflammatory signaling in allergic airway diseases and highlight their potential contribution to the dissemination of type 2 inflammatory responses.
Key Words: Allergic rhinitis; small EVs; IL-13; CCL-26; type 2 inflammation; airway epithelium
1. INTRODUCTION
Extracellular vesicles (EVs) are lipid bilayer–enclosed particles actively released by cells into the extracellular milieu and are now recognized as critical mediators of intercellular communication 1. EVs transport a heterogeneous cargo composed of lipids, nucleic acids, and proteins, including components derived from the plasma membrane and cytosol, as well as proteins involved in metabolic pathways. Early studies considered these vesicles to be cellular byproducts with limited biological relevance. This perspective has since shifted substantially, as EVs are now known to participate in diverse physiological and pathological processes, including immune regulation, cellular homeostasis, and disease progression 2. Despite increasing recognition of their biological importance, the specific contribution of EVs to inflammatory signaling pathways remains insufficiently defined. Given their ability to influence immune responses, elucidating the role of EVs in inflammatory diseases is of particular interest. Notably, their involvement in allergic rhinitis has received minimal attention. A clearer understanding of EV-mediated communication in allergic rhinitis could identify novel therapeutic targets and inform strategies to alleviate symptoms in the large global population affected by this condition. In the present study, EVs within the 30–150 nm size range are referred to as small EVs in accordance with MISEV 2023 guidelines.
Interleukin-13 (IL-13) is a central cytokine involved in type 2 immune responses and plays a prominent role in allergic inflammation 3. Elevated IL-13 signaling is characteristic of airway inflammatory disorders, including asthma and allergic rhinitis. Experimental and clinical studies have demonstrated that activation of the IL-4/IL-13 signaling axis disrupts epithelial barrier integrity in these conditions. Importantly, therapeutic inhibition of IL-4 receptor alpha has been shown to restore epithelial barrier function, underscoring the pathological impact of IL-13–mediated signaling on epithelial homeostasis 3,4. These findings highlight IL-13 as a key driver of epithelial dysfunction and suggest that modulation of IL-13–dependent pathways may reduce inflammatory damage in allergic airway diseases.
CCL-26, a member of the CC chemokine family, is primarily produced by epithelial cells and is strongly induced by IL-13 and IL-4 signaling 5,6. Traditionally, CCL-26 has been characterized as a pro-inflammatory mediator due to its ability to recruit eosinophils and basophils to sites of inflammation 7. However, recent evidence indicates that CCL-26 also contributes to immune resolution by promoting eosinophil clearance 8. This dual role positions CCL-26 as an important regulator of both inflammatory initiation and resolution, emphasizing its relevance in studies investigating the mechanisms governing allergic inflammation.
2. MATERIALS AND METHODS
2.1 Cell Culture and Treatment
Human lung epithelial cells A549 (ATCC- A-549 CCL-185) were used as an in vitro model of airway epithelium. Cells were cultured under standard conditions and monitored regularly until approximately 90% confluency was reached. Upon confluency, cells were assigned to one of three experimental groups: (i) IL-13 treatment (IL-13: PeproTech Thermo Fisher Scientific, Waltham, MA, USA), (ii) vehicle control treated with phosphate-buffered saline (PBS), or (iii) untreated control. Treatments of 100 ng/ml of IL-13 were applied for 24 hours, during which cell morphology and viability were routinely assessed by light microscopy. All experimental procedures were reviewed and approved by the Institutional Biosafety Committee (IBC) prior to study initiation.
2.2 Extracellular Vesicle Isolation
Following treatment, culture supernatants were collected for EV isolation. EVs were isolated using the ExoQuick-TC (Systems Biosciences, Palo Alto, CA, USA) precipitation method according to the manufacturer’s protocol. This approach was selected based on prior reports demonstrating high EV yield and reproducibility compared with ultracentrifugation, which is labor-intensive and resource-demanding 9–11. Briefly, ExoQuick-TC reagent was added to clarified conditioned media and incubated at 4 °C for 30 minutes. Samples were then centrifuged at 13,000 rpm for 2 minutes, after which the supernatant was discarded, and the EV-containing pellet was resuspended and purified using ethanol and wash buffer as recommended.
2.3 EV Characterization
Isolated EVs were characterized using complementary biophysical and molecular approaches, consistent with current EV characterization guidelines 9,12. Particle size distribution and concentration were assessed by nanoparticle tracking analysis (NTA), which quantifies vesicle concentration based on Brownian motion under laser illumination 13. Protein-based validation of EV identity was performed using Western blot analysis. CD63 (Systems Biosciences, Palo Alto, CA, USA) was used as a positive EV marker, while calnexin (Santa Cruz Biotechnology, Dallas, TX, USA) served as a negative control to confirm the absence of cellular contamination 14,15. Concordance between NTA and Western blot results was required to confirm successful EV isolation.
To further evaluate vesicle morphology Transmission electron microscopy (TEM) was performed using a JEOL JEM-2100 transmission electron microscope equipped with a Bruker energy-dispersive spectroscopy (EDS) system. Freshly purified EV samples were negatively stained with uranyl acetate. Briefly, 8 µL of EV suspension was applied to a carbon film–coated TEM grid and incubated for 3 minutes. Excess liquid was carefully removed using filter paper. The grid was then incubated with 2% uranyl acetate for 1 minute, and excess stain was removed using filter paper. Grids were air-dried at room temperature prior to imaging. Samples were examined at an accelerating voltage appropriate for biological specimens, and images were acquired at magnifications corresponding to a 100 nm scale bar to assess vesicle morphology and size distribution.
2.4 EV Cargo Analysis
Protein expression within EVs was assessed using SDS-PAGE followed by Western blotting to quantify levels of the inflammatory chemokine CCL-26. This analysis was performed to determine whether IL-13 stimulation alters the abundance of CCL-26 in EVs. Isolated EV samples were lysed in RIPA buffer containing protease inhibitors, and total protein concentration was determined using a bicinchoninic acid (BCA) assay. Equal amounts of protein were mixed with Laemmli sample buffer under reducing conditions and heated at 95 °C for 5 minutes prior to electrophoresis. Proteins were separated on 4–20% Mini-PROTEAN® TGX™ precast gels (Bio-Rad Laboratories, Hercules, CA, USA) at 100 V for approximately 1 hour. Following separation,proteins were transferred onto polyvinylidene fluoride (PVDF) membranes using the Trans-Blot® Turbo™ Transfer System (Bio-Rad Laboratories, Hercules, CA, USA). Membranes were blocked for 1 hour at room temperature using LI-COR® blocking buffer (LI-COR Biosciences, Lincoln, NE, USA) and incubated overnight at 4 °C with a primary antibody specific for CCL-26 under gentle agitation. After washing three times with Tris-buffered saline containing 0.1% Tween-20 (TBST), membranes were incubated with IRDye®-conjugated secondary antibodies (LI-COR Biosciences, Lincoln, NE, USA) for 1 hour at room temperature. Membranes were washed again and imaged using the Odyssey® Infrared Imaging System (LI-COR Biosciences, Lincoln, NE, USA). Band intensities were quantified using Image Studio software and normalized to total protein staining.
For transcript-level analysis, RNA was isolated from EV samples and reverse-transcribed to complementary DNA (cDNA) prior to quantitative real-time PCR (qPCR). cDNA synthesis was performed using EV RNA combined with polyadenylation reagents and incubated at 37 °C for 30 minutes, following established protocols (Systems Biosciences, Palo Alto, CA, USA) 16. CCL26 mRNA expression was quantified using TaqMan-based quantitative real-time PCR (qPCR) on a QuantStudio™ 5 Real-Time PCR System (Thermo Fisher Scientific, Waltham, MA, USA). qPCR reactions were performed using TaqMan Universal PCR Master Mix and a predesigned TaqMan Gene Expression Assay specific for CCL26 (Thermo Fisher Scientific, Waltham, MA, USA), with a validated housekeeping gene (GAPDH) used for normalization. Reactions were run in technical triplicates with no-template and no–reverse transcriptase controls included to monitor contamination and genomic DNA amplification. Thermal cycling conditions consisted of an initial enzyme activation at 95 °C for 10 min, followed by 40 cycles of denaturation at 95 °C for 15 s and annealing/extension at 60 °C for 1 min. Quantification cycle (Ct) values were obtained using QuantStudio Design and Analysis Software v1.5.
3. RESULTS
3.1 Validation of Extracellular Vesicle Isolation
EVs were successfully isolated from A549 cell culture supernatants. Western blot analysis demonstrated the presence of the EV marker CD63 in EV-positive samples (Figure 1). The absence of calnexin, an endoplasmic reticulum marker, confirmed minimal cellular contamination in the isolated vesicle preparations.

NTA further validated EV isolation (Figure 2). The majority of detected particles fell within the expected size range of 30–150 nm, consistent with small extracellular vesicles. Particle distribution profiles demonstrated a relatively uniform vesicle population, supporting the purity and integrity of the isolated samples.
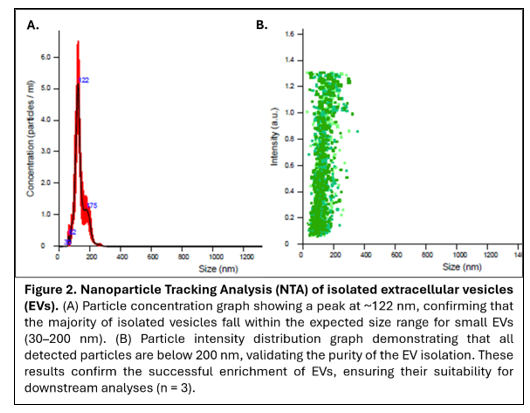
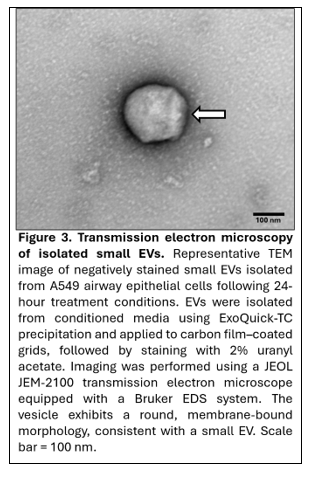
TEM confirmed the presence of EVs in the isolated samples (Figure 3). The vesicles appeared as round to cup-shaped, membrane-bound structures with well-defined lipid bilayers. The majority of particles were within the expected size range of approximately 30–150 nm, consistent with small extracellular vesicles. Minimal background debris or cellular fragments were observed, supporting the purity of the isolated preparations. These morphological findings further corroborate the successful isolation and structural integrity of extracellular vesicles obtained from the experimental groups.
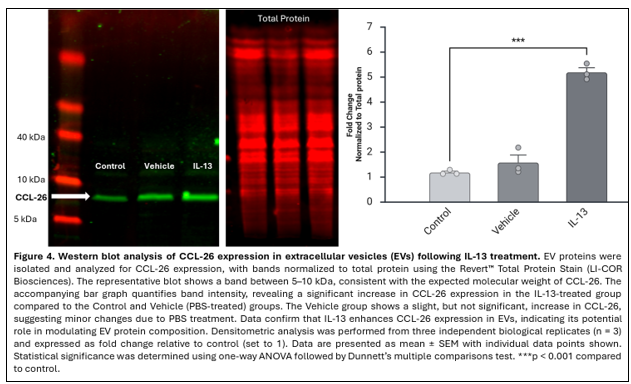
3.2 IL-13 Treatment Increases CCL-26 Protein in EVs
Western blot analysis was performed to assess CCL-26 protein expression within EVs (Figure 4). A band corresponding to the expected molecular weight of CCL-26 (~8 kDa) was identified. Quantitative densitometric analysis was performed from three independent biological replicates (n = 3) and normalized to total protein staining. Protein expression levels were expressed as fold change relative to the control group (set to 1).
Statistical analysis using one-way ANOVA followed by Dunnett’s multiple comparisons test demonstrated a significant increase in CCL-26 protein levels in the IL-13 treatment group compared to control (F(1,4) = 428.367, p < 0.001; adjusted p < 0.0001). Data are presented as mean ± SEM. These results confirm that IL-13 stimulation significantly enhances CCL-26 protein enrichment in EVs.IL-13 stimulation served as the biological positive condition for induction of CCL-26 expression in epithelial cells. Although recombinant CCL-26 protein was not included as a separate positive control in the Western blot analysis, the expected molecular weight (~8 kDa) and IL-13–dependent increase in band intensity were consistent with previously reported observations in epithelial cell models
3.3 CCL-26 mRNA Expression in EVs
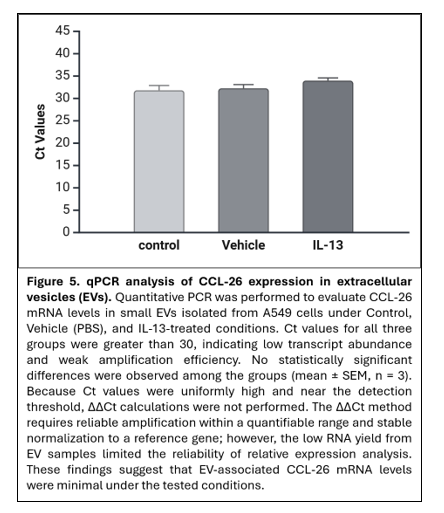
Quantitative real-time PCR was performed to evaluate CCL-26 mRNA levels within EV samples (Figure 5). Ct values for all experimental groups (control, vehicle, and IL-13) were greater than 30, indicating low transcript abundance. No significant differences in Ct values were observed among the three groups.
4. DISCUSSION
Allergic rhinitis remains one of the most prevalent chronic inflammatory disorders worldwide, affecting approximately 40% of the global population 17. Despite its high prevalence, definitive curative therapies remain limited. Improved understanding of the molecular and cellular mechanisms underlying nasal inflammatory diseases may facilitate the development of novel therapeutic strategies. Allergic rhinitis is initiated when the nasal mucosa encounters environmental allergens, leading to activation of T helper 2 (TH2) cells and induction of a type 2 inflammatory response. This immune cascade promotes the release of several cytokines, including interleukin-13 (IL-13), a central mediator of allergic inflammation 3. Clinically, allergic rhinitis manifests as sneezing, nasal congestion, throat irritation, and increased mucus production. Vascular dilation and recruitment of eosinophils, basophils, and helper T cells further amplify the inflammatory response 17.
EVs have emerged as key mediators of intercellular communication. Although historically regarded as cellular byproducts, EVs are now recognized as biologically active carriers of proteins, lipids, and nucleic acids that influence immune signaling and tissue responses 18. EVs are broadly classified based on size and biogenesis; however, current guidelines recommend the use of operational terms such as “small EVs” when biogenesis cannot be definitively determined (MISEV guidelines 2023) 12. Small EVs typically range from 30–150 nm in diameter and are present in multiple biological fluids relevant to airway inflammation, including saliva, bronchial fluid, and nasal secretions 2. Despite increasing recognition of their biological significance, the mechanistic contribution of small EVs to allergic inflammation remains incompletely defined.
Cell-to-cell communication involves the release of signaling molecules, receptor engagement, signal transduction, and downstream cellular responses 19. Within allergic inflammation, IL-13 functions as a principal signaling cytokine. Prior studies have demonstrated that IL-13 stimulation upregulates CCL-26 in epithelial cells. Specifically, IL-13 significantly increases CCL-26 production, whereas, as per Wang et al. 2019, IL-17A alone does not induce comparable changes 20, supporting CCL-26 as a downstream effector of IL-13–mediated inflammatory signaling. A recombinant CCL-26 protein control was not included in the Western blot analysis. While IL-13 stimulation served as a biological positive control, the inclusion of purified CCL-26 protein would further strengthen the validation of band specificity in future studies.
In the present study, successful isolation of small EVs was confirmed through the detection of CD63 and the absence of calnexin, consistent with minimal cellular contamination 14,15. EVs were isolated using ExoQuick precipitation, which efficiently recovers vesicles from conditioned media. However, precipitation-based methods may co-isolate non-vesicular components such as protein aggregates or lipoproteins. In this study, EV identity was supported by complementary characterization approaches, including nanoparticle tracking analysis, transmission electron microscopy, and detection of EV markers (CD63), in the absence of calnexin. Nevertheless, future studies incorporating orthogonal isolation methods, such as size-exclusion chromatography or ultracentrifugation, as well as additional purity metrics, such as particle-to-protein ratios, would further strengthen EV characterization. Nanoparticle tracking analysis demonstrated a predominant vesicle size distribution within the expected 30–150 nm range 2, aligning with current minimal reporting standards for small EV characterization (MISEV 2023) 12.
Protein analysis revealed that IL-13 treatment significantly increased CCL-26 levels within small EVs. The observed band at approximately 8 kDa corresponded to the expected molecular weight of CCL-26 21. Quantitative analysis demonstrated a substantial increase in EV-associated CCL-26 protein in the IL-13 group compared with control conditions. These findings suggest that IL-13 enhances inflammatory protein packaging into small EVs, supporting a potential role for EV-mediated dissemination of type 2 inflammatory signals. Although increased CCL-26 protein levels were observed in EVs following IL-13 stimulation, functional studies were not performed to determine whether these EVs can transfer signals to recipient cells. Future studies investigating EV-mediated effects on immune or epithelial cell responses will be necessary to establish the functional relevance of EV-associated CCL-26 in type 2 inflammation.
In contrast, qPCR analysis revealed no significant differences in CCL-26 mRNA levels among experimental groups. All Ct values exceeded 30, indicating low transcript abundance within small EV samples 22. While this may reflect selective enrichment of protein cargo over mRNA, it may also be influenced by technical limitations associated with low RNA yield from EV preparations. A positive control for EV-associated RNA was not included in the present study, which limits interpretation of these findings. Future studies incorporating EV-enriched RNA controls and more sensitive detection methods will be necessary to distinguish between biological and technical factors. The discrepancy between increased protein levels and unchanged mRNA expression may reflect post-transcriptional regulation, selective protein loading into small EVs, or technical limitations in detecting low-abundance EV-associated RNA. Therefore, conclusions regarding transcriptional regulation of CCL-26 within small EVs remain preliminary.
The study did not assess additional inflammatory mediators such as nerve growth factor (NGF), which may further clarify the role of small EVs in type 2 inflammation. Moreover, optimization of EV RNA extraction and amplification protocols may improve sensitivity in future studies. As an exploratory investigation, these findings suggest that small EVs may contribute to IL-13–mediated inflammatory signaling; however, additional studies are required to determine their functional significance and therapeutic potential. Emerging literature supports continued investigation of EV-mediated modulation of inflammatory responses 23. Another limitation of the present study is the use of the A549 epithelial cell line as an experimental model. Although A549 cells are commonly used to investigate cytokine-mediated airway inflammation, they originate from lung carcinoma and may not fully represent primary nasal epithelial cells involved in allergic rhinitis. Future studies using primary nasal epithelial cells or air–liquid interface culture models would provide a more physiologically relevant system to validate these findings. Future studies should focus on improving small EV RNA detection methodologies, expanding inflammatory cargo profiling, and evaluating functional consequences of EV-mediated CCL-26 transfer. Such investigations may further elucidate the role of small EVs in allergic inflammation and inform the development of targeted therapeutic approaches.
Our findings are consistent with the growing literature supporting a role for epithelial cell-derived EVs in airway inflammation. Prior studies in asthma have shown that IL-13 alters epithelial EV cargo and promotes inflammatory cell responses, including monocyte proliferation and chemotaxis, suggesting that EVs can function as active mediators of type 2 inflammatory communication. Recent data from nasal epithelial EV studies have similarly shown associations between EV cargo and type 2 inflammatory signatures in upper airway disease. In comparison with these reports, the present study identifies increased CCL-26 protein enrichment in EVs following IL-13 stimulation, further supporting the concept that epithelial EVs participate in inflammatory signaling. However, unlike studies emphasizing broader inflammatory EV cargo changes, our data showed increased EV-associated CCL-26 protein without a corresponding increase in EV-associated mRNA, suggesting that selective protein loading or post-transcriptional regulation may contribute to this response. These findings extend prior observations in IL-13–driven airway inflammation while also highlighting the need for future studies to determine whether EV-associated CCL-26 has direct functional effects on recipient epithelial or immune cells 24.
5. CONCLUSION
This study successfully isolated and characterized small EVs from A549 airway epithelial cells. The findings demonstrate that IL-13 treatment increases CCL-26 protein levels within small EVs, supporting a role for EV-associated protein cargo in cytokine-driven type 2 inflammation. Western blot analysis confirmed significant upregulation of CCL-26 at the protein level following IL-13 stimulation. In contrast, qPCR analysis did not reveal significant differences in CCL-26 mRNA levels among experimental groups, limiting conclusions regarding transcriptional regulation within small EVs.
Overall, these results indicate that IL-13–mediated inflammatory stimulation alters EV protein cargo, suggesting a potential role for small EVs in intercellular communication during type 2 inflammation. Further studies are required to clarify the mechanisms governing EV cargo regulation and to determine the broader implications of these findings in inflammatory disease pathogenesis and therapeutic development.
ACKNOWLEDGMENTS
We thank the OSU Microscopy Facility for access to instrumentation and technical support in imaging small EVs. The microscopy assistance provided by Brent Johnson during TEM data acquisition is gratefully acknowledged. The Microscopy Facility at Oklahoma State University provides shared research support and advanced imaging capabilities that were essential to this work
REFERENCES
1. Van Niel G, D’Angelo G, Raposo G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol. 2018;19(4):213-228. doi:10.1038/nrm.2017.125
2. Doyle L, Wang M. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells. 2019;8(7):727. doi:10.3390/cells8070727
3. Nur Husna SM, Md Shukri N, Mohd Ashari NS, Wong KK. IL-4/IL-13 axis as therapeutic targets in allergic rhinitis and asthma. PeerJ. 2022;10:e13444. doi:10.7717/peerj.13444
4. Steelant B, Seys SF, Van Gerven L, et al. Histamine and T helper cytokine-driven epithelial barrier dysfunction in allergic rhinitis. J Allergy Clin Immunol. 2018;141(3):951-963.e8. doi:10.1016/j.jaci.2017.08.039
5. Abonyo BO, Alexander MS, Heiman AS. Autoregulation of CCL26 synthesis and secretion in A549 cells: a possible mechanism by which alveolar epithelial cells modulate airway inflammation. Am J Physiol Lung Cell Mol Physiol. 2005;289(3):L478-488. doi:10.1152/ajplung.00032.2005
6. Macías M, Rebmann V, Mateos B, et al. Comparison of six commercial serum exosome isolation methods suitable for clinical laboratories. Effect in cytokine analysis. Clin Chem Lab Med CCLM. 2019;57(10):1539-1545. doi:10.1515/cclm-2018-1297
7. Shinkai A, Yoshisue H, Koike M, et al. A novel human CC chemokine, eotaxin-3, which is expressed in IL-4-stimulated vascular endothelial cells, exhibits potent activity toward eosinophils. J Immunol Baltim Md 1950. 1999;163(3):1602-1610.
8. Moon HG, Kim SJ, Kim KH, et al. CX3CR1+ Macrophage Facilitates the Resolution of Allergic Lung Inflammation via Interacting CCL26. Am J Respir Crit Care Med. 2023;207(11):1451-1463. doi:10.1164/rccm.202209-1670OC
9. Tang YT, Huang YY, Zheng L, et al. Comparison of isolation methods of exosomes and exosomal RNA from cell culture medium and serum. Int J Mol Med. 2017;40(3):834-844. doi:10.3892/ijmm.2017.3080
10. Cheng J, Wang X, Yuan X, Liu G, Chu Q. Emerging roles of exosome-derived biomarkers in cancer theranostics: messages from novel protein targets. Am J Cancer Res. 2022;12(5):2226-2248.
11. Zhao Z, Wijerathne H, Godwin AK, Soper SA. Isolation and analysis methods of extracellular vesicles (EVs). Extracell Vesicles Circ Nucleic Acids. Published online 2021. doi:10.20517/evcna.2021.07
12. Welsh JA, Goberdhan DCI, O’Driscoll L, et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. J Extracell Vesicles. 2024;13(2):e12404. doi:10.1002/jev2.12404
13. Comfort N, Cai K, Bloomquist TR, Strait MD, Ferrante Jr. AW, Baccarelli AA. Nanoparticle Tracking Analysis for the Quantification and Size Determination of Extracellular Vesicles. J Vis Exp. 2021;(169):62447. doi:10.3791/62447
14. Lässer C, Eldh M, Lötvall J. Isolation and characterization of RNA-containing exosomes. J Vis Exp JoVE. 2012;(59):e3037. doi:10.3791/3037
15. Ivanusic D, Denner J. The large extracellular loop is important for recruiting CD63 to exosomes. MicroPublication Biol. 2023;2023. doi:10.17912/micropub.biology.000842
16. Haque S, Vaiselbuh SR. Exosomes molecular diagnostics: Direct conversion of exosomes into the cDNA for gene amplification by two-step polymerase chain reaction. J Biol Methods. 2018;5(3):e96. doi:10.14440/jbm.2018.249
17. Zhou Y, Chen R, Kong L, Sun Y, Deng J. Neuroimmune communication in allergic rhinitis. Front Neurol. 2023;14:1282130. doi:10.3389/fneur.2023.1282130
18. Lorite P, Domínguez JN, Palomeque T, Torres MI. Extracellular Vesicles: Advanced Tools for Disease Diagnosis, Monitoring, and Therapies. Int J Mol Sci. 2024;26(1):189. doi:10.3390/ijms26010189
19. Su J, Song Y, Zhu Z, et al. Cell–cell communication: new insights and clinical implications. Signal Transduct Target Ther. 2024;9(1):196. doi:10.1038/s41392-024-01888-z
20. Wang WW, Zhu K, Yu HW, Pan YL. Interleukin‐17A potentiates interleukin‐13‒induced eotaxin‐3 production by human nasal epithelial cells from patients with allergic rhinitis. Int Forum Allergy Rhinol. 2019;9(11):1327-1333. doi:10.1002/alr.22382
21. Dunn JLM, Caldwell JM, Ballaban A, Ben-Baruch Morgenstern N, Rochman M, Rothenberg ME. Bidirectional crosstalk between eosinophils and esophageal epithelial cells regulates inflammatory and remodeling processes. Mucosal Immunol. 2021;14(5):1133-1143. doi:10.1038/s41385-021-00400-y
22. Zucha D, Kubista M, Valihrach L. Tutorial: Guidelines for Single-Cell RT-qPCR. Cells. 2021;10(10):2607. doi:10.3390/cells10102607
23. Lou K, Luo H, Jiang X, Feng S. Applications of emerging extracellular vesicles technologies in the treatment of inflammatory diseases. Front Immunol. 2024;15:1364401. doi:10.3389/fimmu.2024.1364401
24. Park KS, Lässer C, Lötvall J. Extracellular vesicles and the lung: from disease pathogenesis to biomarkers and treatments. Physiol Rev. 2025;105(3):1733-1821. doi:10.1152/physrev.00032.2024




